Abstract process experimental period of nearly two years of production and that the fluoroboric acid system to 2,4-- dichlorobenzaldehyde as Additive Sn-Pb alloy plating mirror bright, process stability.
Batch Waste Tyre Pyrolysis Plant
Pyrolysis Plant is used for recycling waste tire, waste plastic, waste rubber to fuel oil. According to the actual situation in every country and district, we developed different models of Batch Pyrolysis Plants for waste tires, rubber and plastic with daily capacity 5 -10 tons.
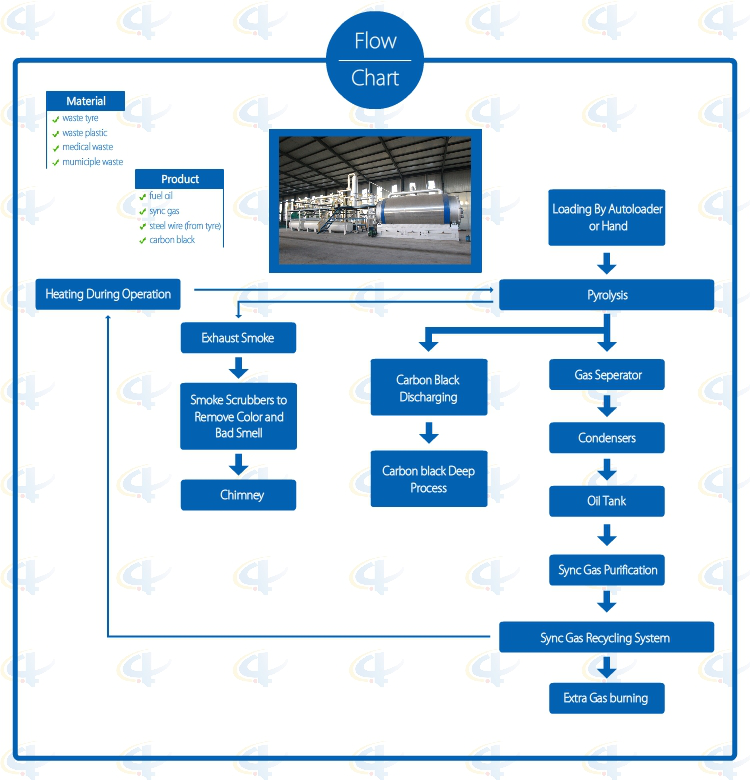
Flowchat of Batch Waste Tyre Pyrolysis Plant
Advantages of Batch Waste Tyre Pyrolysis Plant
1. Full open door design: Speedy loading in and steel pulling out; Easily cooling down after one batch finished, saving time; No leaking with special high temperature flexible graphite packing.
2. Unique Craft Condensers: High condensing efficiency with more oil output. Good quality oil, longer lifetime, and easy to clean.
3. National Patent Unique Smoke Scrubbers: More efficient removal of the acid gas and dust of the smoke by neutralization, purification and absorption, environmental friendly without pollution.
4. National Patent Carbon Black Discharging System: Speedy fully enclosed Auto-discharging under high temperature, avoiding carbon black pollution, saving time.
5. Automatic Submerged welding technology, ultrasonic nondestructive testing, both manual and the automatic safety devices.
6. Sync Gas Recycling System: Fully burned after recycling and utilization, saving fuel and preventing pollution.
7. Direct Heating System: Enlarging heating square to lengthen the lifespan of the reactor and easy to control the temperature.
8. National Patent, unique heat insulation shell; high efficiency temperature keeping, excellent energy-saving effect.
Technical Parameter of Batch Waste Tyre Pyrolysis Plant
NO.
ITEM
PROJECT
1
Equipment Model
XY-7
XY-8
2
Door Model
Full Open Door
Full Open Door
3
Suitable Raw Materials
Rubber/Plastic Products
Rubber/Plastic Products
4
Structure
Horizontal Type Revolves
Horizontal Type Revolves
5
Reactor Size
Φ2200*6000mm Φ2600*6600mm
Φ2200*6000mm Φ2600*6600mm
6
Capacity for One Batch
5-6Mt; 8-10Mt
5-6Mt; 8-10Mt
7
Oil Yield of Tires
40%-45%
40%-45%
8
Work Pressure
Normal Pressure
Normal Pressure
9
Reactor Rotation Speed
0.4R/M
0.4R/M
10
Fuels Choice
Coal, Wood
Coal, Wood, Gas, Oil
11
Power
18KW/H
18-25KW/H
12
Cooling Method
Water Cycling
Water Cycling
13
Type of Drive
External Annular Gear
External Annular Gear
14
Heating Method
Direct
Direct
15
Type of Installation
With Foundation
With Foundation/Integrated Base
16
Noise dB(A)
≦85
≦85
17
Operation Mode
Intermittent Operation
Intermittent Operation
18
Total Weight(MT)
25-40
25-40
19
Installation Space Required
30m*10m
30m*10m
20
Manpower
3~4/batch
3~4/batch
21
Shipment
Ф2200×6000=1*40HC+1*40FR
Ф2600×6600=2*40HC+1*40FR
Ф2200×6000=1*40HC+1*40FR
Ф2600×6600=2*40HC+1*40FR
Batch Waste Tyre Pyrolysis Plant Batch Waste Tyre Pyrolysis Plant,Waste Tyre Pyrolysis Plant,Rubber Pyrolysis Recycling Plant Shangqiu Jinpeng Industrial Co., Ltd. , https://www.recyclings.nl
I products are more silver plated member, but after a period of time is placed silver black, solderability and resistivity will be greatly affected. After DJB-823 is applied, its appearance is affected, and it is necessary to have conductivity under a certain pressure. Therefore, it is imperative to develop a silver-based material.
At that time, many reports that Sn-Ce alloy was a better silver substitute material, we started this process test, but the effect was not satisfactory. So we developed the bright Sn-Pb process, first around the purchase of commercial additives test, but the effect is not ideal, mainly because the coating is not bright, the solution is unstable, so we checked the data and in-depth test, finally found a comparison Good process specifications, after two years of trial production, the performance is quite stable. There are no additives in almost two years, but always keep the high current plating and plating mirror bright. Today I will introduce the work of these years to everyone, I hope everyone can benefit from it. .
1 Process specification
1.1 Process recipe Sn 2+ 18-25g/l
Pb 2+ 8-11g/l
HBF 4 (free) 250-300g/l
H 3 BO 3 : 10-30g/l
OP21 10-30g/l
2.4-dichlorobenzaldehyde lg/l
O-toluidine 6-10ml/l
HCHO 5-15g/l
Dk 5-15A/dm 2
Cathode movement 40-60 times / min
Tem room temperature 1. 2 process
Pretreatment - copper cyanide base - cleaning - plating Sn - Pb - cleaning - passivation - cleaning - drying - inspection [next]
1.3 Precautions 1.3.1 Additives Add 2,4-dichlorobenzaldehyde to o-toluidine for a period of time, then add OP21, HCHO can be added to the bath separately.
1.3.2 The workpiece should not be placed too dense to prevent local fogging.
1.3.3 If the bath has not been used for a long time, test the current density range with HULL before plating.
1.3.4 The coating is bright, so it is supplemented by high-speed cathode movement.
1.3.5 Suitable for iron and copper alloy substrates.
1.3.6 Unqualified plating removal is carried out in the usual way.
2 plating quality inspection
2.1 Appearance The coating is blue with a mirror finish.
2.2 Bonding force according to SJ1282—73 qualified 2.3 third-order performance
Execution according to GJB367, 2-87, wet heat and salt spray test, qualified 2.4 welding performance
Better than silver plating, good fluidity.
3, Conclusion <br> The process has been put into production in 1995, the effect is very good, the coating mirror is bright, and the efficiency is very high, for high-current electroplating, generally plating for 5-10 minutes, from the solution to the present, has not been adjusted once and supplemented The additive proves that the performance of the plating solution is extremely stable. The disadvantage is that the plating solution is a fluoroboric acid system, which has certain requirements for environmental protection, but the process is much better than the many additives at home and abroad in terms of production, which is us. Do a lot of conclusions from comparative experiments.
References 1, Zeng Hualiang, etc., "Electroplating Process Manual" Machinery Industry Press 1993
2, (United States) Frederick "Modern Electroplating" Machinery Industry Press 1982
3. Chen Lizhen, Tsinghua University Science Report, 1979